Intrahepatic cholangiocarcinoma
University of Tsukuba Hospital
Drs. Katsuhiro Nasu and Manabu Minami, Dept. of Diagnostic Radiology, Graduate School of Comprehensive Human Sciences Majors of Medical Sciences
DATE : 2011
- Intrahepatic cholangiocarcinoma and hepatic metastases with associated hepatic infarction
- Information about diagnosis of intrahepatic cholangiocarcinoma and peritoneal dissemination obtained by Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI with this patient
- Information about diagnosis of hepatic micrometastases from intrahepatic cholangiocarcinoma obtained by Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI with this patient.
Intrahepatic cholangiocarcinoma and hepatic metastases with associated hepatic infarction

Patient’s background and objectives of MRI
The patient, a male in his 50s, was treated for repeated upper abdominal pain at a local hospital, and an infarction was found in the left lobe of the liver, so he was referred to the authors’ hospital. However, he had no underlying diseases likely to lead to hepatic infarction, and hepatic infarction also seemed unlikely on the basis of his clinical progression, so thorough tests were performed, taking into consideration the possibility that a malignant tumor was present as an underlying disease.
Slice section 1
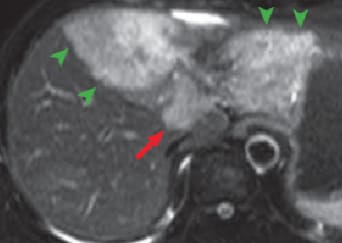
(a) T2-weighted image

(b) Diffusion-weighted image
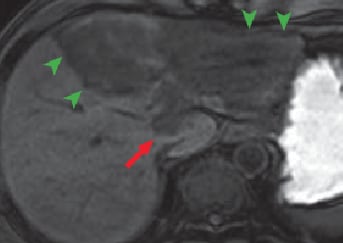
(c) Pre-contrast T1-weighted image
MRI
Except for the caudate lobe, all left lobes of the liver showed high signals with T2-weighted imaging (a), and diffusion-weighted imaging (b), and with pre-contrast T1-weighted imaging (c) the region was observed as having an inhomogeneously low signal (green arrows). In addition, with diffusion-weighted imaging, a high-signal region was found at the liver surface (blue arrows).

(d) Arterial phase
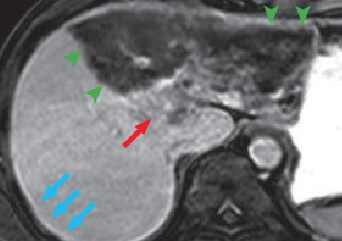
(e) 180 s after contrast agent administration
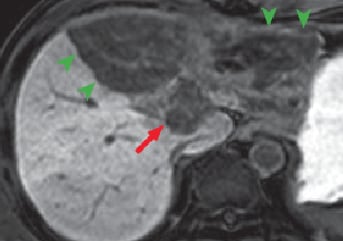
(f) Hepatobiliary phase
Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI
Except for the region close to the portal region, all left liver lobes showed low signals, in all phases, with no effective contrast, and hepatic cirrhosis was confirmed (green arrows). In the portal region of the left lobe, in the arterial phase (d) imaging of the nodule showed gradually increasing enhancing effect for 180 s after contrast agent administration (e; red arrows). These contrast effects with the nodule became unclear in the hepatobiliary phase (f). The findings strongly suggested intrahepatic cholangiocarcinoma. In addition, during the arterial phase and for 180 s after contrast agent administration, the findings were consistent with those with diffusion-weighted imaging, with a high-signal region at the liver surface (blue arrows).
Slice section 2
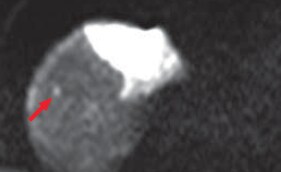
(a) Diffusion-weighted image
MRI
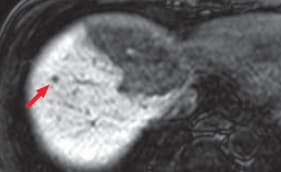
(b) Hepatobiliary phase
Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI
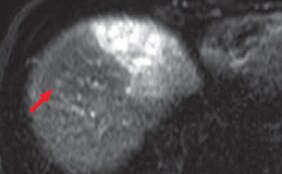
(c) T2-weighted image
MRI
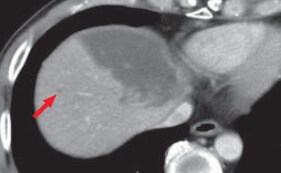
(d) Portal phase
MDCT
Hepatic metastases were clearly shown by diffusion-weighted imaging (a) and in the hepatobiliary phase (b; red arrows). It is considered that, as long as the existence of lesions can be recognized, with T2-weighted imaging (c) and in the portal phase of computed tomography (d), both of which are reliable, it can be demonstrated that the lesions are small nodules rather than cysts or hemangiomas.
Information about diagnosis of intrahepatic cholangiocarcinoma and peritoneal dissemination obtained by Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI with this patient
Nodules shown by red arrows are signs strongly suggesting intrahepatic cholangiocarcinoma. However, the tumor-enhancing effects became unclear in the hepatobiliary phase (f). On the basis of decrease in blood concentration, it is considered probable that Gadoxetate disodium(Gd-EOB-DTPA) distributed in the intralesional extracellular fluid was removed. This suggests that a different theory about diagnosis is needed from the one accepted to date, as the current theory is that diseases such as hemangioma and intrahepatic cholangiocarcinoma are diagnosed making use of the property of the available extracellular fluid contrast agents being distributed solely in the extracellular fluid, which is one of their most important characteristics.
Hepatic cirrhosis (green arrow) is thought to develop due to constriction of the hepatic artery and portal vein by the tumor. Of course, uptake by hepatic parenchyma that has become cirrhotic cannot be visualized in the hepatobiliary phase, even with contrast effects in a dynamic study.
On the other hand, the region with the blue arrow is strongly suspected of showing peritoneal dissemination. In MRI using extracellular fluid contrast agents, if diffusion-weighted imaging shows a high signal at the hepatic surface (more accurately, the right subphrenic space), when delayed enhancement effects are confirmed in the same region, peritoneal dissemination was diagnosed. However, this diagnostic criterion is probably not applicable when Gadoxetate disodium(Gd-EOB-DTPA) is used. Due to the strong enhancement effects in the lesion and the neighboring hepatic tissue, whether or not contrast is shown is difficult to determine. In the case of the present patient, with imaging up to 180 s after contrast agent administration in Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI, it can be confirmed that disseminated lesions show faint contrast, but it is rather difficult to identify the relevant signs without diffusion-weighted imaging. Unless it is sufficiently recognized that Gadoxetate disodium(Gd-EOB-DTPA) is no more than a hepatocellular-carcinoma-specific contrast agent, with the property of being an extracellular fluid contrast agent, there is the risk of missing extrahepatic lesions that used to be diagnosable. An additional issue is that, although the present patient’s peritoneal dissemination could not be clearly demonstrated pathologically, disseminated lesions were definitely present in the Morrison’s pouch and pouch of Douglas, and there is certainly no doubt that the findings in the right subphrenic space represented dissemination.
Information about diagnosis of hepatic micrometastases from intrahepatic cholangiocarcinoma obtained by Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI with this patient.
The present patient was also suspected of having intrahepatic metastases, and the final treatment selected at the cancer-specialist hospital was chemotherapy. The presence of intrahepatic metastases can of course be suggested even by the portal phase of computed tomography alone. However, if the issue is how to achieve fully reliable diagnosis using computed tomography alone, the authors consider that confirmation should be made by means of diffusion-weighted imaging and in the hepatobiliary phase. Another point is that a disadvantage that has been found with superparamagnetic iron oxide (SPIO) contrast MRI is its high sensitivity for hepatic metastases combined with difficulty of distinguishing small metastases from tangential sections of intrahepatic blood vessels.
The hepatobiliary phase of Gadoxetate disodium(Gd-EOB-DTPA) contrast MRI gives much higher image quality than SPIO contrast MRI, and also enables thin-slice imaging, so discrimination from blood vessels is considered to be easier than with SPIO contrast MRI. However, when separation of vessels and micrometastases is readily achievable by means involving sequencing, there is no better approach than that. It is therefore considered that it will continue to be necessary to use diffusion-weighted imaging for intra- and extrahepatic evaluation even after introduction of Gadoxetate disodium(Gd-EOB-DTPA).
- *The case introduced is just one clinical case, so the results are not the same as for all cases.
- *Please refer to the Package Insert for the effects and indications, dosage and administration method, and warnings, contraindications, and other precautions with use.


